Title
A comparative trial of a novel flow limiting device attached to a manual ventilation bag versus a manual ventilation bag alone during forceful breaths in patients undergoing non-emergent surgery with general anesthesia: A prospective crossover randomized controlled trial.
B. Rapsas, MD; K. Dankievitch, BS; E. Lee, MD; M. Brady, MD, MPH; M. Kendall, MD
Presented at:
-
American Society of Anesthesiologists (ASA) Annual Meeting 2025
-
European Resuscitation Council (ERC) Congress 2025
A prospective randomized crossover trial evaluated a novel flow-limiting device attached to a manual ventilation bag compared with a manual ventilation bag alone during forceful breaths in patients undergoing non-emergent surgery under general anesthesia.
Background
Poor manual ventilation can result in excessive tidal volumes and high airway pressures, regardless of provider experience. These conditions increase the risk of gastric insufflation and aspiration.
In 2022, the FDA approved a flow-limiting device (Sotair®, SafeBVM Corp.) designed to restrict flow rates to ≤55 L/min, with the goal of reducing excessive airway pressures and gastric insufflation.
Objective
The study aimed to compare peak airway pressures and tidal volumes during forceful manual ventilation with and without the flow-limiting device in patients undergoing general anesthesia for non-emergent surgical procedures.
Methods
-
Design: Randomized two-group crossover superiority trial
-
Population: Adult ASA I–III patients undergoing non-emergent surgery under general anesthesia
-
Protocol: After endotracheal intubation, patients received forceful manual ventilations every 30 seconds for 3 minutes with and without the flow-limiting device, in randomized order
-
Definition of forceful ventilation: Breaths that triggered the APL (pop-off) valve set at 35 cm H₂O
Primary endpoint:
Mean peak inspiratory pressure (PIP) from five forceful breaths per condition.
Hypothesis:
Use of the flow-limiting device would reduce mean PIP by 7.5 ± 7.5 cm H₂O compared with manual ventilation alone.
Statistical analysis:
Generalized mixed effects modeling.
Patient Characteristics
-
Total patients: 30
-
Sex: 12 male (40%), 18 female (60%)
-
Age: 47.9 ± 13.8 years
-
BMI: 28.9 ± 5.3 kg/m²
Results
A total of 150 forceful breaths were analyzed.
Peak Flow Rate
-
Higher during manual ventilation without the device
-
Reduced when the flow-limiting device was used
Peak Airway Pressure
-
Higher without the device
-
Lower with the flow-limiting device
Tidal Volume
-
Higher without the device
-
Lower when the flow-limiting device was used
All comparisons between groups were statistically significant (p < 0.001).
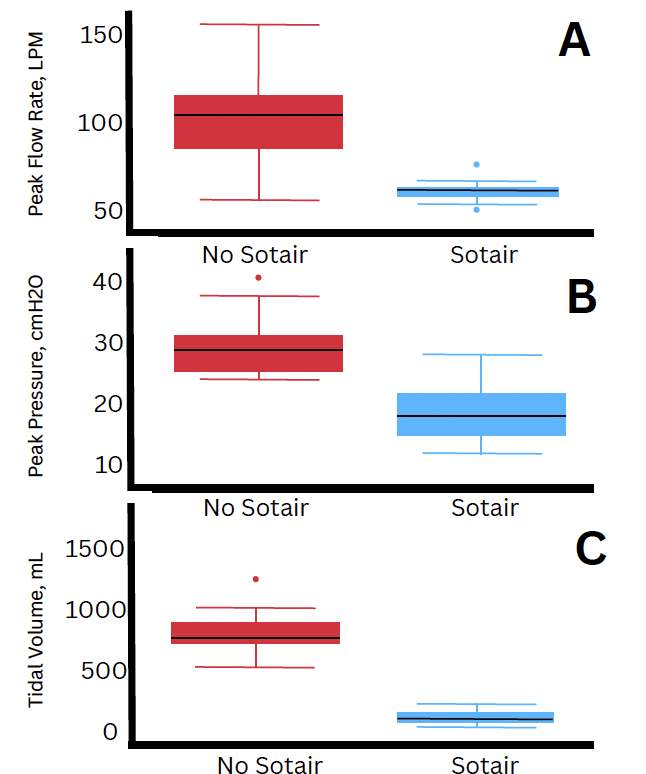
during “forceful” ventilations. These ventilation events were designed to test the impact of
flow limitation on overventilation as measured via a Sensirion SFM3300-D disposable airflow
sensor. By design, a flexible diaphragm in the Sotair completely obstructs airflow through the
airway circuit when 55LPM is exceeded. Panel A shows that Peak Airway Flow, the
instantaneous maximum in the airway flow waveform, was vastly lower in the flow-limited
group, and in practice reflects a transient spike above the design limit of 55LPM. Panel B
demonstrates the range of pressures under flow limitation was markedly lower as well. Panel
C highlights the consequence of complete flow-gating: a potential overventilation event
becomes a non-ventilation. Mean peak presure, tidal volume and peak flow rate differed
between groups p (<0.001).manual ventilation bag alone during forceful breaths in patients undergoing non-emergent surgery
with general anesthesia: A prospective crossover randomized controlled trial.
Conclusion
Adding the flow-limiting device to a manual ventilation bag reduced peak airway pressures and excessive tidal volumes during forceful breaths compared with manual ventilation alone.
References: 1) J Clin Monit Comput. 2020:34, 771-77 2) Resuscitation. 2005:64(3) 321-5. 3) https://www.safebvm.com/

